Catalysis : theories

source : slide share.net
Theories of Catalysis-
There are two theories to explain the mechanism of action of a particular catalyst.
1) Intermediate compound theory
2) Adsorption Theory
Intermediate Compound Theory
This theory is used mainly to explain the mechanism of homogeneous catalysis let us consider the following general reaction
A + B ——> AB
A & B are reactants & X is a Catalyst & AB is the product . According to this theory one of the reactant first react with the catalyst & forms an intermediate compound , which is relatively less stable & reacts with another reactant to form the product & give back the catalyst.
A+ X ——>AX (Intermediate Compound)
AX + B ——> AB + X
product catalyst
Ex.
1 : When ethyl alcohol (excess) is heated with conc. H2SO4 at 1400C, diethyl ether, is obtained H2SO4 acts as a catalyst in this reaction.
2 C2H5OH——–> C2H5–O–C2H5+H2O
Ethoxy Ethane
Mechanism-
C2H5OH + H2SO4(conc.) ——-> C2H5HSO4 (Intermediate ) + H2O
C2H5HSO4 + C2H5OH——–> C2H5–O–C2H5 + H2SO4 (Catalyst)
Ex. 2 : The Catalytic oxidation of SO2 to SO3 in lead chamber process in presence of NO catalyst takes place as-
2SO2 + O2——> 2SO3
Mechanism-
2NO + O2 ——> 2NO2
Catalyst Nitrogen dioxide (Intermediate compd)
NO2 + SO2 ( Reactant ) ——-> SO3 + NO ( Catalyst)
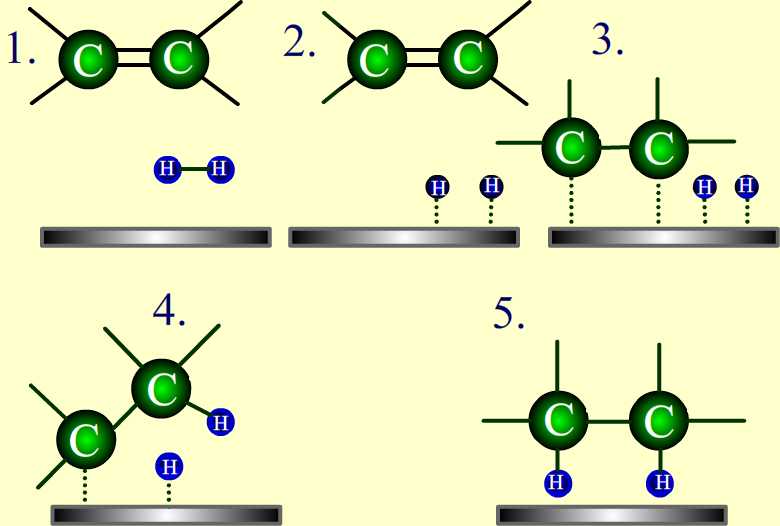
Adsorption Theory-
The accumulation of a substance on the surface of another substance is known as adsorption.
The substance which is accumulated on the surface of another substance is known as adsorbate, The compound which accumulates another substance on its surface is known as adsorbant. Absorbants are usually solids.
The Mechanism involves the steps-
1. Diffusion of reactants to the surface of the catalyst
2. Association between the catalyst surface & the reactants occur. It means adsorption takes place.
3. Occurence of chemical reaction on the catalyst surface.
4. Desorption of reaction products from the catalyst surface.
5. Diffusion of reaction products away from the catalyst surface
Free Valencies-
Activity of catalyst is due to the presence of free valencies . More is the free valencies at the surface of the catalyst, greater will be its activity. The no. of free valencies can be increased by subdivision of the solid & by increasing the roughness of the surface of the solid. Fine powdered or colloidal catalyst particles having large surface area .Hence very rich in free valencies. The surface of the solid is not smooth. It has peaks, corners & cracks. Greater is the roughness of the surface, greater is no of peaks & cracks. Peaks, cracks & corners present at the surface of the catalyst are known as active centres. Greater is the no. of active centres , more effective will be catalyst.
Facts explained by Adsorption theory-
1. Activity of Collodial Catalyst-
Surface area of sub divided & colloidal catalyst is very large therefore the number of free valencies at the surface of the catalyst is very high So the catalyst is very effective catalyst.
2. Action of Catalyst promoters-
Calalytic promoter increases the roughness of the surface of the catalyst therefore the number of active centres & the free valencies are increased. Hence catalyst become more effective.
3. Action of Catalytic Poisons-
Free valencies are present at the surface of the catalyst. Thus poisons combine with the free valencies present at the surface therefore number of free valencies become less . Hence the activity of the catalyst is low.
4. The surface of the Catalyst is used again & again due to alternate adsorbtion & desorption hence small quantity of catalyst can catalyse large amount of reactants.
5. Desorption leads the catalytic surface unchanged therefore the catalyst at the end of reaction remains same in mass & composition.