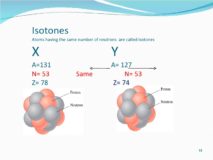
Isotones
isotones source : www.youtube.com ISOTONES Term isotone was given by German Physicist Guggen heimer. Definition Atoms of different Continue Reading »

De Broglie Equation
De Broglie equation source :http://switkes.chemistry.ucsc.edu According to De Broglie, a moving small particle like Continue Reading »

Isobars
isobars source : academics easy.com Atoms of different elements having same mass number but different atomic number, are Continue Reading »
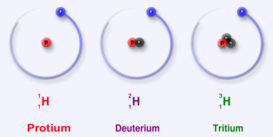
Isotopes
isotopes source :quara.com Isotopes were first discovered by Soddy in radioactive elements while isotopes of nonradioactive elements were Continue Reading »

Heisenberg’s Uncertainty Principle
source : science20.com Heisenberg Principle “It is impossible to measure simultaneously the position and momentum or velocity Continue Reading »

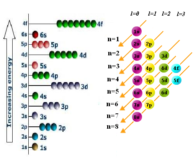
(n+l) Rule-sequence of filling subshell
subshell source : knowledge bin.org Subshell (n+l) rule The Sequence of the filling of various sub-shells may also Continue Reading »
Aufbau Principle-
Aufbau principle source : slide player.com Aufbau (German word) means ‘building up’ according to Aufbau principle, “The vacant sub Continue Reading »
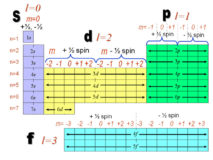
Numericals of Quantum Numbers-
Quantum number source : physics.tutor vista.com Quantum numbers Question1: What is value of Principal & Azimuthal Quantum Number of Continue Reading »
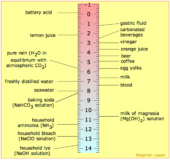
pH (Power of Hydrogen ion)
Hydrogen ion concentration source : youtube.com Hydrogen ion concentration- Sorenson gave a new term pH to express hydrogen ion Continue Reading »

Ostwald Dilution Law
Ostwald dilution law source : slide player.com Ostwald dilution law is an application of law of mass action Continue Reading »