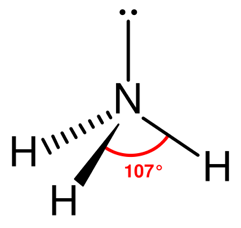
Ammonia
source : story boardthat.com
Method of preparation: Ammonia
1. By the reaction of water with metal nitride:
Mg3N2 (Magnesium nitride + 6H2O ——-> 3Mg(OH)2 +2NH3
ALN (Aluminium nitride) + 3H2O ——–> Al(OH)3+ NH3
2. By heating ammonium salt with base:
2NH4Cl + Ca(OH)2 ——-> CaCl2 +2NH3 +2H2O
2NH4Cl + PbO ——-> PbCl2 + 2NH3+ H2O
NH4Cl + NaOH ——-> NaCl + NH3 + H2O
Lab method :
In laboratory, NH3 is prepared by heating ammonium chloride with calcium hydroxide. Concentrated H2SO4, anhydrous CaCl2, P2O5 cannot be used to dry NH3 because they react chemically with NH3.

source : you tube.com
H2SO4 (Concentrated) +2NH3 ———> (NH4)2SO4
CaCl2 +8NH3 ———-> CaCl2.8NH3
P2O5 + 3H2O +6NH3 —————–>2 (NH4)3PO4
Manufacture:
It is manufactured by three method:
1) From ammonical liquor:
Ammonical liquor is obtained as a by product when coal gas is manufactured by the destructive distillation of coal. Ammonical liquor contains ammonium salt such as NH4Cl, (NH4)2SO4, (NH4)2S etc. Ammonia is obtained by heating ammonical liquor with slaked lime (Ca(OH)2 ). Ammonia obtained is either liquefied or dissolved in dilute H2SO4 to get ammonium sulphate which is used as fertilizer.
2NH4Cl + Ca(OH)2 ———>CaCl2 + 2NH3 + 2H2O
(NH4)2SO4 + Ca(OH)2 ———>CaSO4 + 2NH3 + 2H2O
(NH4)2S + Ca(OH)2 ———>CaS + 2NH3 + 2H2O
2NH3 + H2SO4 (dilute) ———> (NH4)2SO4
Physical properties:
1. It is colorless, pungent odor gas.
2. Ammonia is highly soluble in H2O. Its aqueous solution is called ammonium hydroxide.
NH3 +H2O ——-> NH4OH (weak base)
Test of Ammonia :
1. Ammonia has pungent ammonical odor.
2. Its aqueous solution turn red litmus blue.
3. Copper sulphate solution react with excess of NH4OH then blue color soluble complex is obtained.
4. Concentrated HCl gives white fumes of NH4Cl with NH3 .
5. It gives orange-brown precipitate with alkaline solution of K2[HgI4] i.e Nessler’s reagent
2K2[HgI4] + NaOH + NH3 ———> NH2Hg2I3 (brown precipitate) + 4 KI + NaI +H2O
NH2Hg2I3 is Iodide of million’s base.