General Chemistry
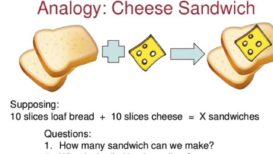
Stoichiometry numerical -2
Stoichiometry numerical Stoichiometry numerical- Question 1) How many moles of N2 are needed to produce 8.2 moles of NH3 Continue Reading »

Stoichiometry numerical -1
Stoichiometry numerical- Question- 23 gm of sodium reacts with water. Calculate (i) the weight of H2 liberated (ii) the Continue Reading »

Stoichiometry
Stoichiometry Stoichiometry- In a reaction, quantitative relationships among the reactants & products is called ‘Stoichiometry’. Stoicheion means element & Continue Reading »

Percentage composition
Percentage composition- Percentage composition gives the mass of each element expressed as the percentage of the total mass. It Continue Reading »
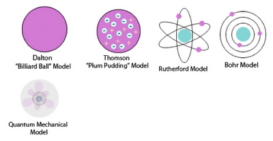
Dalton atomic theory
Dalton atomic theory To provide justification to the laws of chemical combination, Dalton proposed atomic theory. The basic postulates Continue Reading »
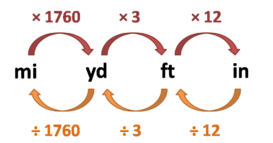
Unit conversion
Unit conversion- Unit conversion – One unit can be converted into another by conversion factor method or dimensional analysis. Continue Reading »

Properties of matter
Properties of matter- Properties of matter- Each substance has unique properties, these are physical & chemical properties. Physical properties Continue Reading »
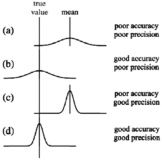
Accuracy and precision
Accuracy and precision- source: mathsis fun.com Accuracy and precision- Accuracy- Accuracy means the closeness of an experimental value or Continue Reading »

Scientific notation
Scientific notation- Scientific notation- A better and simplified way to represent very large or very small values is called Continue Reading »
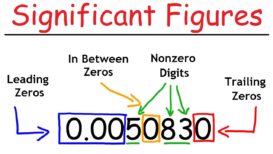
Significant figures
Significant figures- Significant figures- “The significant figures of a number are digits that carry meaningful contributions to its measurement Continue Reading »