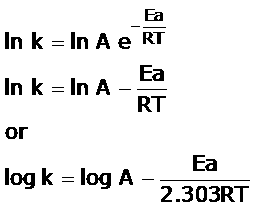Arrhenius equation-

source: Slideshare.net
Arrhenius equation-
Q 1) The slope of line in the graph of ‘log K ‘ Vs ‘1/T’ for a reaction is -5400 K. Calculate the energy of activation ?
(R = 8.314 JK-1Mole-1)
Solution )
Slope = -5400 K ; Ea = ? ; R = 8.314 JK-1Mole-1
According to Arrhenius equation,
slope = -Ea / 2.303 R
-5400 = – Ea / 2.303 x 8.314
Ea = 103394.57 J mole-1
Ea = 103.39 KJ mole-1 Ans.
Q 2) The rate constant of a first order reaction becomes 6 times when the temperature is raised from 350 K to 410 K .
Calculate the energy of activation ? (R = 8.314 JK-1Mole-1)
Solution )
T1 = 350 K ; T2 = 410 K ; K2 = 6K1
According to Arrhenius equation,
log [K2 / k1] = [Ea/ 2.303 R][(1 / T1) – (1 / T2)]
log [6K1 / k1] = [Ea/ 2.303 x 8.314 ][(1 / 350) – (1 / 410)]
log 6 = [Ea/ 2.303 x 8.314 ][(410 -350) / (350 x 410)]
0.7782 =Ea x 60 / 2.303 x 8.314 x 410 x 350
Ea = 35636.56 J mole-1
Ea = 35.64 KJ mole-1 Ans.
Q 3 ) In the Arrhenius equation for a reaction , the value of A and Ea are 4 x 10 13 sec-1 and 98.6 K J mole-1 respectively. If the reaction is of first order , at what temperature will it half life period be 10 minutes ?
Solution )
t 1/2= 10 minute = 10 x 60 sec = 600 sec ; A = 4 x 10 13 sec-1 ; Ea = 98.6 K J mole-1 = 98.6 x 103 J mole-1
K = 0.693 / t1/2 = 0.693 / 600
K = 1.155 x 10-3 sec-1
According to Arrhenius equation,
log K = log A – Ea / 2.303 RT
log (1.155 x 10 -3) = log (4 x 10 13) – [98.6 x 10-3 / 2.303 x 8.314 x T]
log 1.155 +l og 10 -3 = log 4 + log 10 13 – [98.6 x 10 3 / 2.303 x 8.314 x T]
log 1.155 – 3l og 10 = log 4 + 13 log 10 – [98.6 x 10 3 / 2.303 x 8.314 x T]
0.06258 – 3 x 1 = 0.6020 + 13 x 1 – [ 98.6 x 10 3 / 2.303 x 8.314 x T]
– 2.9374 = 13.6020 – 5149.59 / T
– 2.9374 – 13.6020 = -5149.59 / T
16.5394 = 5149.59 / T
T = 311.35 K Ans.
Q 4 ) The rate constant of a reaction is 1.2 x 10 -3 sec-1 at 303 K and 2.1 x 10 -3 sec-1 at 313 K .Calculate the energy of activation?
( R = 8.314 J K -1 mole-1)
Solution )
T1 = 303 K ; T2 = 313 K ; K1 = 1.2 x 10 -3 ; K2 = 2.1 x 10 -3
According to Arrhenius equation,
log [K2 / k1] = [Ea/ 2.303 R][(1 / T1) – (1 / T2)]
log [ 2.1 x 10-3 / 1.2 x 10-3] = [Ea/ 2.303 x 8.314 ][(1 / 303) – (1 / 313)]
(log 2.1 log 1.2) = [Ea/ 2.303 x 8.314 ][(313 -303) / (303 x 313)]
0.3222 – 0.0792 =Ea x 10 / (19.15 x 303 x 313)
Ea = 0.243 x 19.15 x 303 x 313 /10
Ea = 44132.8 J mole-1
Ea = 44.13 KJ mole-1 Ans.
Q 5) The rate of a reaction triples when temperature changes from 200 C to 500 C . Calculate the energy of activation ? (R = 8.314 JK-1Mole-1)
Solution )
T1 = 20 + 273 = 293 K ; T2 = 50 + 273 = 323 K ; K2 = 3 K1
According to Arrhenius equation,
log [K2 / k1] = [Ea/ 2.303 R][(1 / T1) – (1 / T2)]
log [3 K1 / k1] = [Ea/ 2.303 x 8.314 ][(1 / 293) – (1 / 323)]
log 3 = [Ea/ 2.303 x 8.314 ][(323 -293) / (323 x 293)]
0.4771 =Ea x 30 / 2.303 x 8.314 x 293 x 323
Ea = 28817.89 J mole-1
Ea = 28.82 KJ mole-1 Ans.
Q 6 ) The reaction 2NO2 ——-> 2 NO + O2 has an Ea of 110 K J mole-1. At 4000C the rate constant is
7.8 mole -1 litre sec -1. Calculate rate constant at 4300 C ?
Solution )
T1 = 400 + 273 = 673 K ; T2 = 430 + 273 = 703 K ; K2 = ? ; K1 = 7.8 ; Ea = 110 K J mole-1 = 110 x 10 3 J mole-1
log [K2 / k1] = [Ea/ 2.303 R][(1 / T1) – (1 / T2)]
log [ K2 / 7.8 ] = [110 x 10 3/ 2.303 x 8.314 ][(1 / 673) – (1 / 703)]
log (K2 / 7.8) = [110 x 10 3 / 2.303 x 8.314 ][(703 -673) / (703 x 673)]
log (K2 / 7.8) = 0.3643
Taking antilog of RHS,
K2 / 7.8 = antilog 0.3643
K2 / 7.8 = 2.314
K2 = 7.8 x 2.314
K2 = 18.05 mole-1 litre sec-1 Ans.
Q 7 ) A first order reaction is 50 % complete in 30 minutes at 27 0C and in 10 minutes at 470C .Calculate Ea of reaction ?
Solution )
At 270C ,
t 1/2 = 30 min
K1 = 0.693 / t 1/2 = 0.693 / 30
K1 = 0.0231 min-1
At 47 0 C ,
t 1/2 = 10 min
K2 = 0.693 / t 1/2 = 0.693 / 10
K2 = 0.0693 min-1
T1 = 27 + 273 = 300 K ; T2 = 47 + 273 = 320 K ;
log [K2 / k1] = [Ea/ 2.303 R][(1 / T1) – (1 / T2)]
log [0.0693 / 0.0231] = [Ea/ 2.303 x 8.314 ][(1 / 300) – (1 / 320)]
log 3 = [Ea/ 2.303 x 8.314 ][(320 -300) / (320 x 300)]
0.4771 =Ea x 20 / 2.303 x 8.314 x 320 x 300
Ea = 43848.49 J mole-1
Ea = 43.85 KJ mole-1 Ans.