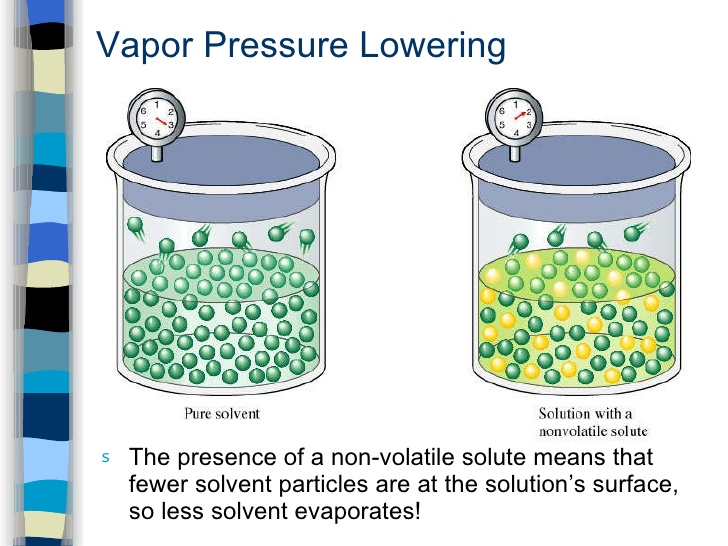
Raoult’s Law

source : slideplayer.com
Raoult’s Law-Numerical
Question 1) Cholesterol is very soluble in ether. If 0.869 gm of cholesterol is dissolved in 4.4 gm of ether (C4 H10O),the vapour pressure of ether lowers from 0.526 to 0.507 atmosphere at 180 C . What is molecular weight of cholesterol ?
Solution )
po= 0.526 atm
Ps = 0.507 atm
po-ps =0.019
w = 0.869 gm
M of ether (C4H10O) = 12 x 4 + 1 x 10 + 1 x 16 = 74
m of cholesterol = ?
W= 4.4 gm.
(po-ps) /ps =wM/Wm
0.019 /0.507 =0.869 x 74 / 4.4 m
m = 0.869 x 74 x 0.507 /0.019 x 4.4
m = 389.99 Ans.
Question 2) The vapour pressure of an aqueous solution of glucose at 373 K is 750 mm Hg. Calculate the molality and mole fraction of solute ?
Solution )
T =373 K
Po of water = 1 atmosphere = 760 mm
Ps = 750 mm
M of water (H2O) = 1 x 2 + 1 x 16 = 18
According to Raoult’s Law
(po-ps) / po =wM / Wm
Molality = w x 1000/ m W
multiply by ‘M’ in numerator & denominator,
Molality = w x 1000 x M / m x W x M
because ,
(po-ps ) /po = wM/Wm
so,
Molality = (po-ps ) x 1000 /po x M
= (760 – 750) x 1000 / 760 x 18
Molality = 0.731 mole / Kg
mole fraction of solute ,
(n /n +N )= (po-ps ) /po
= (760 – 750)/760 = 10 / 760 =0.013
mole fraction of solute = 0.013 Ans.
Question 3 ) Calculate the relative lowering in vapour pressure if 10 gm of a solute ( molecular weight =100) are dissolved in 180 gm water ?
Solution)
According to Raoult’s Law
Relative lowering in vapour pressure ,
po – ps / po = (n /n +N )
w =10 gm., m = 100, W = 180 gm. , M of water = 18
n = w/m = 10 / 100 =0.1
N = W/M= 180 /18 = 10
po – ps / po = 0.1 / (0.1 + 10)
= 0.1 /10.1
[ po – ps / po ] = 0.0099
Relative lowering in vapour pressure = 0.0099 Ans.
Question 4) At 500C the vapour pressure of water and ethyl alcohol are 92.5 & 219.9 mm of Hg respectively . If 6.0 gm of nonvolatile solute (molecular weight = 120) are dissolved in 150 gm of each of these solvent. What will be the ratio of relative vapour pressure lowering in two solvents ?
Solution)
Relative lowering in vapour pressure,
po – ps / po = (n /n +N )
For water,
w =6.0 gm., m = 120, W = 150 gm. , M of water = 18
n = w/m = 6 / 120 =0.0.05
N = W/M= 150 /18 = 8.33
po – ps / po = 0.05 / (0.05+ 8.33 )
Relative lowering in vapour pressure for water = [ po – ps / po] = 0.0059
For ethyl alcohol ,
w =6.0 gm., m = 120, W = 150 gm. , M of ethyl alcohol ( C2H5OH) = 2 x 12 + 5 x 1 + 16 + 1 = 46
n = w/m = 6 / 120 =0.0.05
N = W/M= 150 /46 = 3.26
po – ps / po = 0.05 / (0.05+ 3.26 )
po – ps / po = 0.015