Abnormal colligative properties-

source : Emaze
Abnormal colligative properties-
Dilute solutions containing non volatile solute exhibits some special properties which depend only upon the number of solute particles present in the solution irrespective of their nature .The properties are called colligative properties .The colligative properties are –
a) Lowering of vapour pressure
b) Elevation in boiling point
c) Depression in freezing point
d) Osmotic pressure
These properties are related to one another .If one is measured , the other can be calculated. The results are excellent if three conditions are satisfied –
i) The solution should be very dilute.
ii) The solute should be nonvolatile .
iii) The solute does not dissociate or associate in solution.
The experimental values of colligative properties of nonvolatile , non electrolyte solute in solution resemble to the calculated values of colligative properties . However , in case of electrolyte solute in solution the experimental values of colligative properties are different . Such values are known as abnormal values of colligative properties.
The abnormal behaviour of colligative properties has been observed in the following cases-
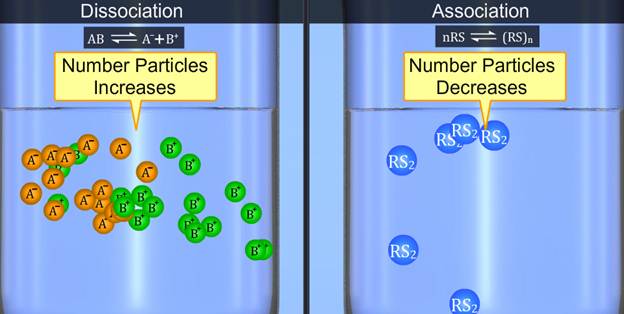
If association of solute occurs-
In case of association , no. of particles in solution decreases Hence experimental value of Colligative properties becomes lower.
Experimental colligative properties < normal colligative properties
Experimental value of lowering in vapour pressure , elevation in boiling point , depression in freezing point and osmotic pressure becomes less than normal values .But experimental value of molecular weight will be greater than normal molecular weight because,
m ∝ 1 / colligative properties
consider , a solute having association nature,
nA ⇌ [A] n
mole before association 1 0
moles after association 1 – ∝ ∝ /n
∝ is degree of dissociation
n = no. of molecules associate
Normal colligative property is proportional to the no. of particles before association.
Normal colligative property = 1.
Experimental colligative property is proportional to the no. of particles after association.
Experimental colligative property = 1 – ∝ + ∝ /n
i= Experimental colligative property / normal colligative property
i =[ 1 – ∝ + ∝ /n] / 1 = normal molecular weight / Experimental molecular weight
i = Vant Hoff factor
i < 1
If dissociation of solute occurs-
Solutes (electrolyte) which dissociate in solvent , show an increase in number of particles present in solution. So this gives to the higher experimental values of colligative property because colligative property depends upon the no. of solute particles only.
Experimental colligative properties > normal colligative properties
Experimental value of lowering in vapour pressure , elevation in boiling point , depression in freezing point and osmotic pressure becomes more than normal values .But experimental value of molecular weight will be less than normal molecular weight because,
m ∝ 1 / colligative properties
Experimental molecular weight < normal molecular weight
consider , a solute having dissociation nature,
A x B y ⇌ x A + + y B –
mole before dissociation 1 0 0
moles after dissociation 1 – ∝ x.∝ y .∝
∝ is degree of dissociation
n = no. of molecules associate
Normal colligative property is proportional to the no. of particles before association.
Normal colligative property = 1.
Experimental colligative property is proportional to the no. of particles after association.
Experimental colligative property = 1 – ∝ + x.∝ + y.∝
i= Experimental colligative property / normal colligative property
i =[ 1 – ∝ + x.∝ + y.∝] / 1 = normal molecular weight / Experimental molecular weight
i = Vant Hoff factor
i > 1

source : Try Not Laughs