d-block elements

source : slide player.com
Elements of d block :
The elements of III B, IV B, V B, VI B, VII B, VIII , VIII ,VIII , I B & II B groups are present in the middle part of periodic table. Now a days these are elements of 3 to 12 group in the periodic table. In d-block elements last electron enters in (n-1)d orbital. These are also called transition elements.
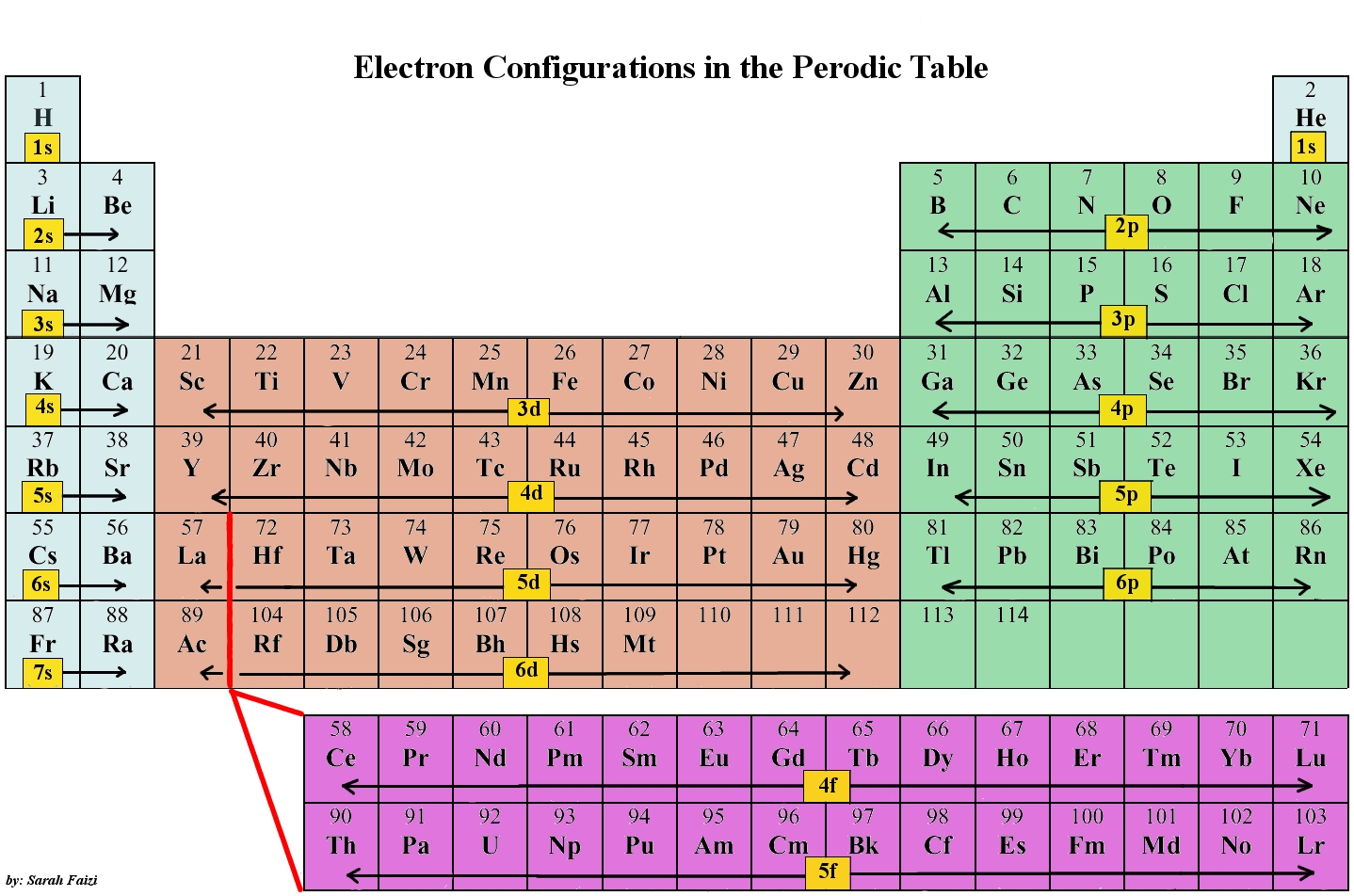
There are 4 series of d-block elements.
3d-series: 21Sc to 30Zn ( Ist transition series i.e the last electron comes in 3d orbital)
4d-series: 39 Y to 48Cd ( II transition series i.e the last electrons comes in 4d orbital)
5d-series: 57 La & 72Hf to 80Hg ( III transition series i.e the last electrons comes in 5d orbital)
6d-series: 89Ac & 104Rf – 112Uub ( IV transition series i.e the last electrons comes in 6d orbital )
General Characteristic of d-block elements :
1) Electronic configuration :
Electronic configuration of outermost shell is:
![]()
& penultimate shell is:
![]()

2) Variable valency : d-block elements show variable valency because electrons present in ns and (n-1)d take part in bond formation .
Valency of Cu is 1 & 2 [cuprous & cupric ]
Valency of Fe is 2 & 3 [ferrous & ferric]
Valency of Au is 1 & 3 [aurous & auric ]
3) Metallic nature : All d-block elements are metals.
4) formation of colored ions : Cations having incompletely filled d-orbitals form colored ions.
Example: Fe++, Fe+++, Cu++, Ni++ etc.
Cations having completely filled d-orbitals form colorless ions.
Example: Cu+, Ag+, Au+, Zn++ etc.
5) Para & diamagnetic character : Atoms & ions containing unpaired electrons are paramagnetic.
Example: Fe, Fe++, Fe+++, Ni++, Cu++ etc.
Atoms & ions in which all the electrons are paired, are diamagnetic.
Example : Zn , Zn++, Cu+, Ag+ etc.
6) Tendency to form complexes : Transition metal cations form complexes due to small size of cation & presence of vacant d-orbitals of appropriate energy.
7) Catalytic property : Elements of d-block and their compounds are used as catalyst.
Example : Fe, Co, Ni, V2O5 , Fe2O3 ,Pt etc.
Read more articles at chemistryonline.guru