Ionic Radius
source : slide player.com
Ionic Radius:
It is equal to the distance between the centre of the nucleus of the ion and its outermost shell.
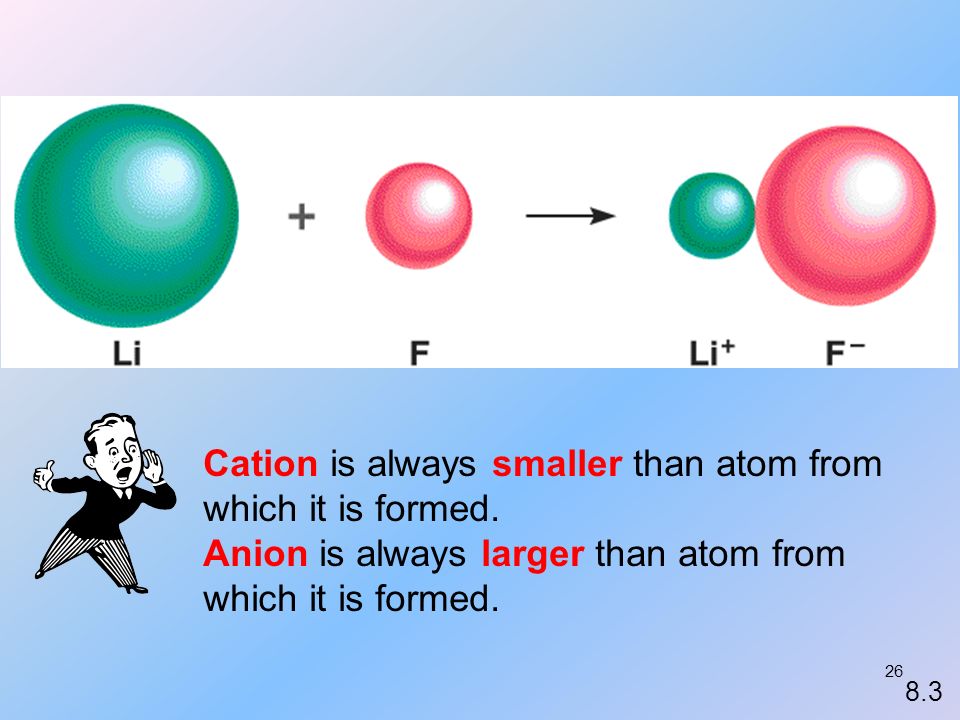
Radius of Cation: radius of cation is always smaller than its neutral atom.
![]()
Reason:
- When a cation is formed usually outermost shell of electrons disappear.
- When electrons are given out the effective nuclear charge of cation is increased.Therefore electrons of outermost shell are attracted towards the nucleus by greater force.

Z/e = 11/10 12/10 13/10
Z/e = 1.1 1.2 1.3
Greater is the Z/e ,smaller is the radius.

Z/e = 26/26 26/24 26/23
Z/e = 1.0 1.08 1.13
Greater is the Z/e ,smaller is the radius.
Radius of Anion:
It is always greater than its neutral atom.
![]()
Reason:
- When one or more electrons are taken up by an atom then effective nuclear charge decreases.
- Hence electrons of the outer most shell are attracted towards the nucleus by less force.
- Thus radius of anion increases.

Z/e = 7/10 8/10 9/10
Z/e = 0.7 0.8 0.9
Greater is the Z/e ,smaller is the radius.

Z/e = 16/18 17/18 20/18
Z/e = 0.89 0.94 1.1
Greater is the Z/e ,smaller is the radius.

Z/e = 8/10 9/10 10/10
Z/e = 0.8 0.9 1.0
Greater is the Z/e ,smaller is the radius.
Variation in atomic radius of elements:
Variation in a group:
Atomic radius of elements increases on moving down the group due to presence of extra shell of electrons.
Variation in a period:
On moving in a period from left to right, atomic radius decreases from alkali metals to halogens . It is because nuclear charge increases but number of shells remain the same . Due to increase in nuclear charge electrons of outer most shell are attracted towards the nucleus by greater force.Therefore atomic radius decreases in a period with increase in atomic number.Radius of inert gases is exceptionally greater because their crystal radius is taken.
Ionization Potential :
“It is equal to the energy required to remove an electron from the outermost shell of an isolated gaseous atom.” Its unit is eV/atom or KJ/gm-atom. It is represented by I.
Question: The value of second Ionization potential of an element is always greater than first Ionization potential why ?
Solution :
After the removal of one electron from the neural atom, the effective nuclear charge of the cation is increased . Therefore electrons of outermost shell of the cation are attracted towards the nucleus by greater force. Hence it becomes difficult to remove the second electron from the outermost shell of cation. Hence greater amount of energy is required to remove second electron . So second Ionization potential of an element is always greater than first Ionization potential .
Similarly Ionization potential third is greater than second Ionization potential .

Read more articles at chemistryonline.guru