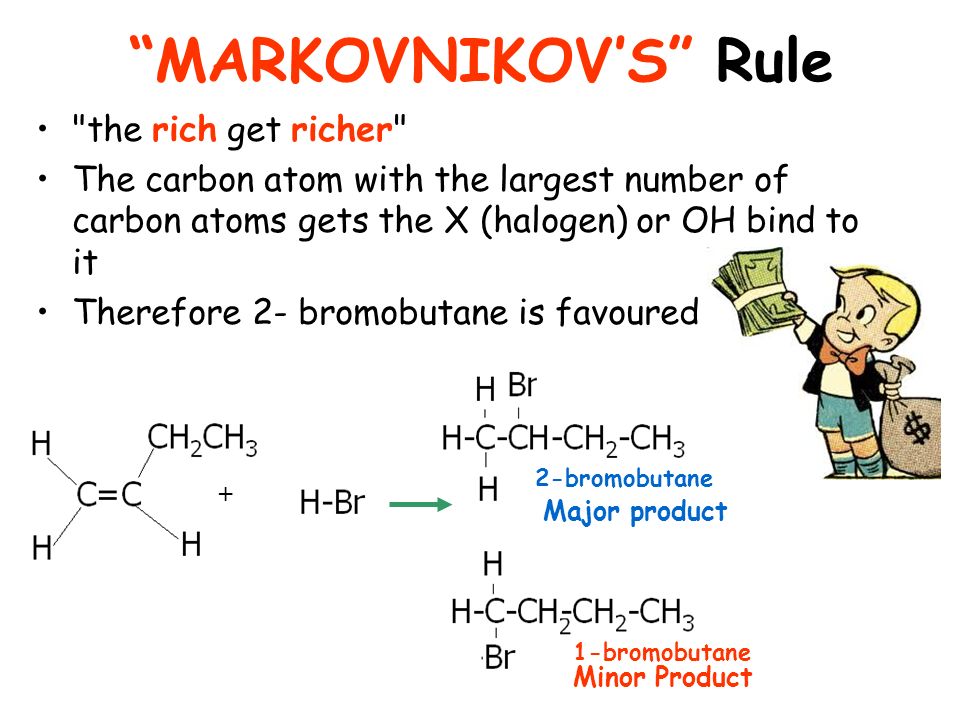Markownikoff rule

source : slideplayer
MarkowniKoff ‘s rule (M.K.Rule)-
“When an unsymmetrical reagent adds to an unsymmetrical alkene, the negative part of the reagent get attached to that carbon atom which is joined to lesser number of Hydrogen atom”.
Ex.1 ) Addition of Halogen acid to Propene :

Ex,2 ) Addition of Halogen acid to alkynes :

Anti Markownikoff’s rule or Kharasch effect or peroxide effect-
The addition of any polar molecule to any unsymmetrical alkene or unsymmetrical alkyne in the presence of oxygen or benzoyl peroxide takes place against the M.K. rule . According to this rule –
When any polar molecule is added to any unsymmetrical alkene in presence of any organic peroxide, the negative part of molecule is added to that carbon atom which is connected to more Hydrogen atom than the other unsaturated carbon atom . This is known as peroxide effect.
Ex. Addition of Halogen acid to Propene :