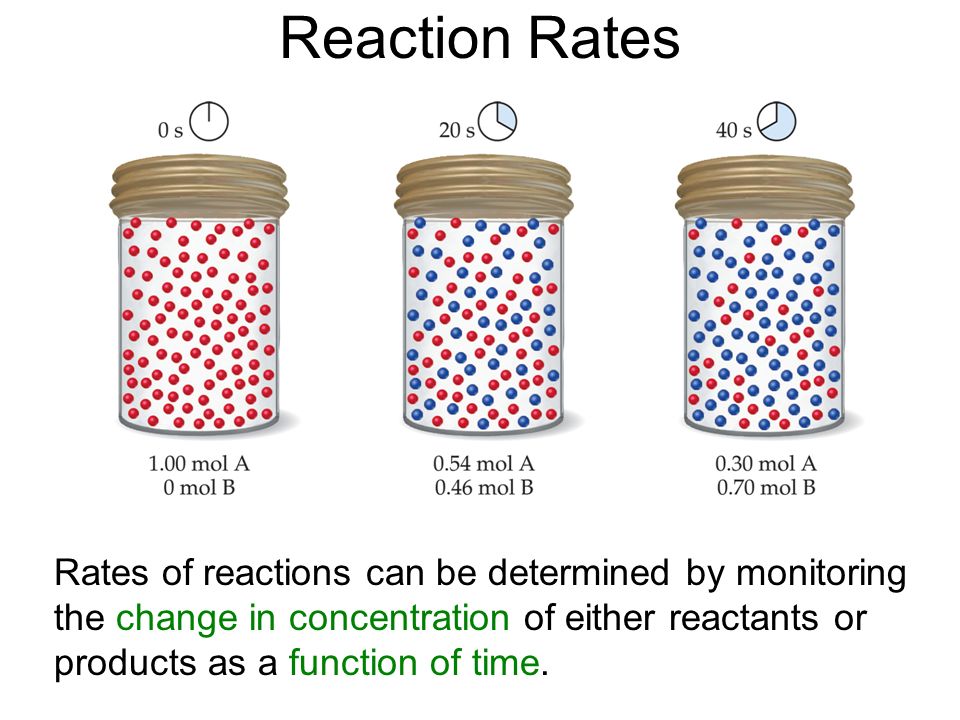
Order of reaction
source : chemguide.co.uk
Order of reaction-
Q 1)
(i) The reaction . A + 3B ———> 2C , obeys the rate equation ,
Rate = K [A]1/2 [B]3/2. Find order of reaction ?
(ii) The reaction A + B —–> C has zero order . Write rate equation ?
Solution )
(i) Rate = dx / dt = K [A] 1/2 [B]3/2
order of reaction = 1/2 + 3/2 =2
Reaction is second order.
(ii) If reaction is zero order then,
Rate = K [A]0[B]0 Ans.
Q 2) Find order of reaction and unit of rate constant-
(i) 2 H2 + 2 NO ——–> 2 H2O + N2
Rate = K [H2][NO]2
(ii) H2O2 + 3 I– + 2 H+ ——–> 2 H2O + I3– (aq.)
Rate = K [H2O2][I–]
(iii) CH3CHO ———> CH4 + CO
Rate = K [CH3CHO]3/2
Solution)
(i) Rate = K [H2][NO]2
Order of reaction = 1 + 2 = 3
Reaction is third order.
Unit of K –
Rate = K [H2][NO]2
K = Rate / [H2][NO]2
unit of K = (mole litre-1 sec-1) / (mole litre-1 )(mole litre-1 )2
Unit of K = litre2 mole -2sec-1 Ans.
(ii)
Rate = K [H2O2][I–]
Order of reaction = 1 + 1 = 2
Reaction is second order.
Unit of K-
Rate = K [H2O2][I–]
K = Rate / [H2O2][I–]
unit of K = (mole litre-1 sec-1) / (mole litre-1 )(mole litre-1 )
Unit of K = litre mole-1 sec-1 Ans.
(iii)
Rate = K [CH3CHO] 3/2
Order of reaction = 3/2
Unit of K-
Rate = K [CH3CHO]3/2
K = Rate / [CH3CHO]3/2
unit of K = (mole litre-1 sec-1) / (mole litre-1 )3/2
Unit of K = litre1/2 mole -1/2 sec-1 Ans.
Q 3) -The rate of reaction,
2A + B ——–> A2B has the rate law,
Rate = K[A]2 with rate constant equal to 0.50 mole -1 litre sec-1. Calculate the rate of reaction when,
(i) [A] = 0.60 mole / litre , [B] = 0.05 mole/litre
(ii) When conc. of A and B have been reduced to 1/4.
Solution)
(i) K = 0.50 mole -1 litre sec-1 , [A] = 0.60 mole /litre
Rate of reaction = K[A]2 = 0.50 x 0.60 x 0.60
Rate of reaction = 0.18 Ans.
(ii)
K = 0.50 mole -1 litre sec-1 , [A] = 0.60 x 1/4 mole /litre
Rate of reaction = K[A]2
= 0.50 x 0.60 x 0.60/ 4 x 4
Rate of reaction = 0.011 Ans.
Q 4) The reaction , 2NO + O2 ——> 2 NO2 follows the rate law,
Rate = K [NO]2[O2]. If K = 2 x 10 -6 litre2 mole-2sec -1. Calculate rate of reaction when [NO] = 0.04 mole /litre and
[O2] = 0.2 mole / litre.
Solution )
[NO] = 0.04 mole /litre , [O2] = 0.2 mole / litre , K = 2 x 10 -6 litre2 mole-2sec -1.
Rate = K [NO]2[O2] = 2 x 10 -6 [0.04]2 [0.2]
Rate =6.4 x 10 -10 mole litre-1 sec-1 Ans.