Rules-IUPAC

source:youtube
source: slide share.net
(A) Rules-IUPAC of Saturated hydrocarbons-
i) Longest chain Rule
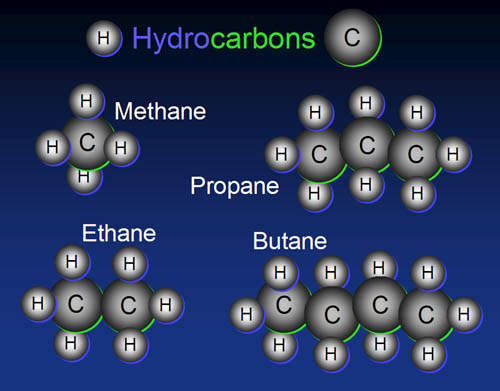
a) The 1st step is to select the longest continuous chain of carbon atoms Which is called parent chain or main chain. Other carbon chains attached to it are known as side chains. On the basis of number of Carbon atoms present in the parent chain, parent hydrocarbon is named.(Root word+ane)
EX-

Longest chain of 5 C-atom is selected as parent chain.

Straight chain contains 6 C – atoms while the longest selected chain contains 8 C- atoms.

Longest selected chain contains 7 C- atoms.
b) When molecule contains two chains of the same length then chain which carries maximum number of side chains is selected as parent chain.
EX – 1)

( Correct )

( Wrong )
EX – 2)

(Correct )

(Wrong )
EX-3)
(Correct )
ii) Numbering of the Carbon-atom of the longest chain
a) The Carbon atoms of the parent chain are numbered by arabic numerals 1,2,3,4 etc from one end to other. The number that locates the position of substituent is known as ‘locant’. The position of side chains are indicated by number assigned to the carbon atom to which it is attached.
b) When many substituents are present, then numbering is done from that end of chain where sum of substituent is minimum .
c) If different alkyl groups are present as substituents on the identical positions in relation to the ends of the chain, then minimum number is given to the smaller alkyl group.







iii) Naming of the Side chains-
a) The name of the substituent is prefixed, preceded by the locant to the name of parent hydrocarbon.
locant,hyphen,substituent,parent hydrocarbon

b) If two or more substituents are present, then each substituent prefixed by its locant, in alphabetical order preceding the name of parent hydrocarbon.
 `
`
c) When the same substituent is present two, three or four times then prefix di, tri or tetra is prefixed before the name of the substituent. The locants are written in increasing order & separated by commas.
The prefixes di, tri, or tetra should not be considered while arranging the substituent alphabetically

d) When the substituent has itself branched chain then it is named as substituted alkyl group. Its chain is also numbered by greek letters or Arabic numbers (1,2,3) from the carbon atom attached to the main chain. The name of such substituent is written in bracket.



source: youtube