Avogadro hypothesis

source: Tutorvista
Avogadro hypothesis-
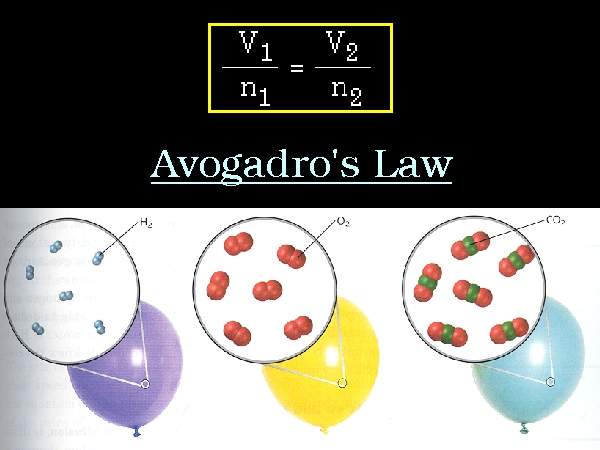
” Under similar conditions of temperature and pressure , equal volumes of all gases contain equal number of molecules.”
V ∝ N
‘N’ is the number of molecules.
Ex –
We have equal volumes of three gases hydrogen (H2) , oxygen (O2) and chlorine (Cl2) in different flasks of the same capacity under similar conditions of pressure and temperature. We find that all the flasks have same number of molecules. These molecules may differ in size and mass.
2H2(g) + O2(g) ———->2H2O(g)
2 mols 1 mol 2 mols
N2(g) + 3H2(g) ———->2NH3(g)
1 mols 3 mols 2 mols
Avogadro hypothesis explains successfully the formation of HCl,
Hydrogen + Chlorine = Hydrogen chloride
Avogadro’s law 1 vol 1 vol 2 vol
n molecules n molecules 2n molecules
1 molecule 1 molecule 2 molecules
1/2 molecule 1/2 molecule 1 molecule
1 atom 1 atom 1 molecule
Hydrogen and chlorine are diatomic in nature. One molecule of Hydrogen chloride contains 1/2 molecule of hydrogen and 1/2 molecule of chlorine.Thus one molecule of Hydrogen chloride is formed from 1 atom of hydrogen and 1 atom of chlorine.
Applications of Avogadro hypothesis-
The important applications of Avogadro’s hypothesis are as follows-
i)Relationship between molecular weight and vapour density-
vapour density = density of gas / density of hydrogen
= mass of certain volume of gas/ mass of same volume of hydrogen at same temp. and pressure
= mass of n molecules of gas / mass of n molecules of hydrogen
= mass of 1 molecules of gas / mass of 1 molecules of hydrogen
= molecular mass of gas / molecular mass of hydrogen
Because molecular mass of hydrogen =2
Thus,
vapour density= molecular mass / 2
Mol. mass = 2 x vapour density
ii) Atomicity –
Number of atoms present in one molecule is called atomicity . Avogadro’s hypothesis is helpful in the determination of atomicity.
Ex – Hydrogen , chlorine ,nitrogen are diatomic while ozone is triatomic and inert gases are monoatomic in nature.
iii) Molecular formula –
This hypothesis is helpful in finding the molecular formula of gases.
iv) Gram molecular volume –
One gram mole of any gas occupies 22400 ml. or 22.4 litre of volume at NTP. According to Avogadro’s hypothesis , equal volumes of different gases contain same number of molecules under similar conditions of pressure and temperature.

source: writeopinions.com