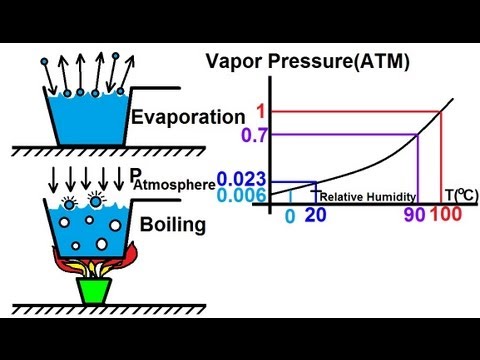
Elevation of boiling point

source : HyperPhysics Concepts
Elevation of boiling point-Numerical
Question 1 ) What will be the boiling point of solution when 174.5 mg of octa atomic sulphur is added to 78 gm of bromine . Kb for bromine is 5.2 K Kg /mole. Boiling point of bromine is 332.15 K.
Solution )
Kb = 5.2 K Kg / mole ,
boiling point of solution = ?
m of sulphur ( octaatomic ) = 32 x 8 = 256
w = 174.5 mg. = 174.5 /1000 = 0.1745 gm.
W = 78 gm
ΔTb =1000 Kb. w /W.m
ΔTb = 1000 x 5.2 x 0.1745 / 78 x 256
ΔTb = 0.045
Elevation in boiling point (ΔTb ) = boiling point of solution – boiling point of pure solvent
boiling point of solvent ( benzene ) = 332.15 K
boiling point of solution = ?
0.045 = boiling point of solution – 332.15
boiling point of solution =332.195 K Ans.
Question 2) The molal elevation constant for water is 0.510 K Kg/mole. Calculate boiling point of solution made by dissolving 6 gm urea in 200 gm water ?
Solution )
Kb = 0.510 K Kg / mole , boiling point of solution = ?
m of urea ( NH2CONH2 ) = 14 + 2 + 12 + 16 +14 +2 = 60
w = 6.0 gm.
W = 200 gm
ΔTb =1000 Kb. w /W.m
ΔTb = 1000 x 0.510 x 6.0 / 200 x 60
ΔTb = 0.255
Elevation in boiling point (ΔTb ) = boiling point of solution – boiling point of pure solvent
boiling point of solvent ( water) = 1000C
boiling point of solution = ?
0.255 = boiling point of solution – 100
boiling point of solution = 100.255 0C Ans.
Question 3) A solution of an organic compound freezes at 272.33 K and boils at 373.187 K. Calculate molal elevation constant for water ? If molal depression constant for water is 1.86 K Kg/mole.
Solution )
freezing point of pure solvent (water ) = 00C = 0 + 273 = 273 K
freezing point of solution = 272.33 K
ΔTf = freezing point of pure solvent – freezing point of solution
ΔTf = 273 – 272.33
ΔTf = 0.67
Elevation in boiling point (ΔTb ) = boiling point of solution – boiling point of pure solvent
boiling point of solvent ( water) = 1000C =100 + 273 =373 K
boiling point of solution = 373.187 K
ΔTb = 373.187 – 373
ΔTb = 0.187
ΔTb /ΔTf = Kb/Kf
Kf = 1.86 K Kg /mole
0.187 / 0.67 = Kb / 1.86
Kb = 0.187 x 1.86 /0.67
Kb = 0.519 = 0.52 K Kg /mole Ans.
Question 4 ) . An aqueous solution of urea has a boiling point 100.180C. Calculate its freezing point ? Molal constants of water Kf and Kb are 1.86 & 0.512 K Kg/mole respectively
Solution )
freezing point of pure solvent (water ) = 00C
freezing point of solution = ?
Elevation in boiling point (ΔTb ) = boiling point of solution – boiling point of pure solvent
boiling point of solvent ( water) = 100 0C
boiling point of solution = 100.18 0C
ΔTb = 100.18 – 100
ΔTb = 0.18
ΔTb /ΔTf = Kb /Kf
Kf = 1.86 K Kg /mole
Kb = 0.512 K Kg /mole Ans.
0.18 / ΔTf = 0.512 / 1.86
ΔTf = 0.654
ΔTf = freezing point of pure solvent – freezing point of solution
0.654 = 0 – freezing point of solution
freezing point of solution = 0 – 0.654
freezing point of solution = – 0.654 Ans