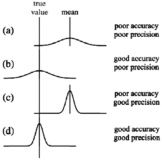
Accuracy and precision
Accuracy and precision- source: mathsis fun.com Accuracy and precision- Accuracy- Accuracy means the closeness of an experimental value or Continue Reading »

Scientific notation
Scientific notation- Scientific notation- A better and simplified way to represent very large or very small values is called Continue Reading »
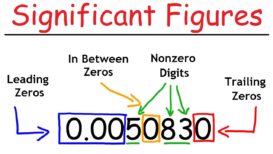
Significant figures
Significant figures- Significant figures- “The significant figures of a number are digits that carry meaningful contributions to its measurement Continue Reading »

Significant figures calculation
Significant figures Calculations involving Significant figures- Rule 1- When addition & subtraction of digits is carried out, the Continue Reading »
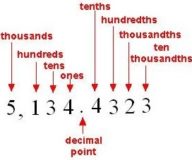
Rounding off the Numbers
Rounding off the Numbers Rounding off the Numbers- Round to the Nearest Hundred Ex- 1650 i) Identify the Continue Reading »
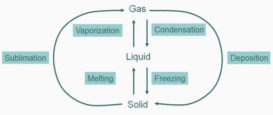
Classification of matter
Classification of matter- Classification of matter- MATTER– It occupies space & possesses mass, it can exist in three Continue Reading »

Rank the following atoms and molecules based on which would effuse the fastest. i) H2 ii) He iii) N2 iv) CO2 v) H2S
Question – Rank the following atoms and molecules based on which would effuse the fastest. i) H2 ii) He Continue Reading »
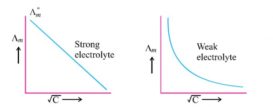
Kohlrausch law- Applications
Kohlrausch law- source : slideplayer.com Applications of Kohlrausch law- 1) Calculation of degree of ionisation and ionisation constant- Degree Continue Reading »

Find the solubility of MnS in neutral water in gm/l .Ksp of MnS = 2.6 x 10-14
Find the solubility of MnS in neutral water in gm/l ? Ksp of MnS = 2.6 x 10-14. Solution Continue Reading »
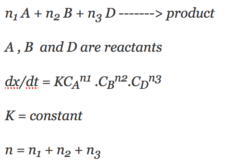
If the initial concentration of cyclopropane is 0.0501 M. What will the cyclopropane concentration after 352minutes.The reaction is first order. Rate constant = 3.36 x 10-5 sec-1.
Question – If the initial concentration of cyclopropane is 0.0501 M. What will the cyclopropane concentration after 352 minutes. Continue Reading »